This article covers
What is plastic?
Plastics are complex polymers, consisting of long and repeating chains of molecules. Plastic is light, durable, and transparent; however, it creates detrimental problems for our environment. It takes more than decades or even hundreds of years to decompose naturally. It sounds horrible, doesn’t it?

[In this figure] Plastic wastes take hundreds of years to break down.
Photo source: futurism.com
Among the plastics, polyethylene terephthalate (PET) is the most commonly used thermoplastic polymer to make plastic products such as polyester clothing, plastic bottles, and food container.
Currently, it is very difficult to break down plastic bottles into their building block to make new ones from old ones. Therefore, new plastic will be made from oil each year.
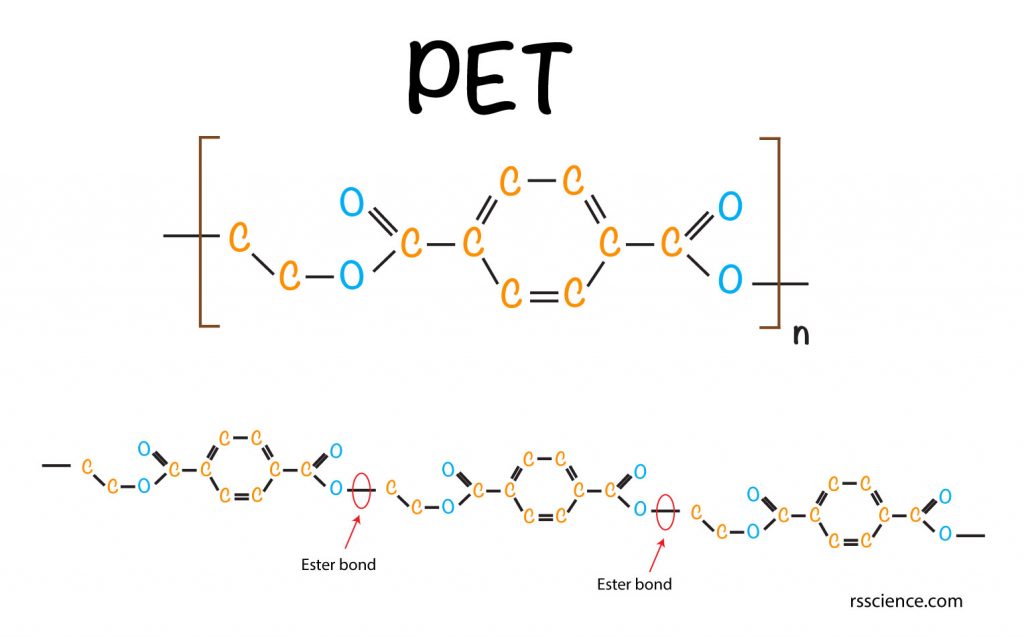
[In this figure] The chemical structure of polyethylene terephthalate (PET).
PET is a polymer consisting of repetitive monomers. N stands for the number of repeats. Individual monomers connect with an ester bond (C-O) with each other, indicated by red arrows.
What are plastic eating bacteria?
Bacteria that can use plastic as their major energy and carbon source for growth called plastic-eating bacteria.
The discovery – where are plastic-eating bacteria found?
In the March 11, 2016 issue of Science, a team of researchers led by Kohei Oda of Kyoto Institute of Technology and Kenji Miyamoto of Keio University reported that they isolated a novel bacterium, Ideonella sakaiensis 201-F6, from a PET contaminated environment near a plastic bottle recycling site among 250 samples.
Ideonella sakaiensis belongs to the genus Ideonella. Its name comes from Sakai, a city in Japan, where the bacterium was found. When the Japanese team cultured Ideonella sakaiensis with PET film, they found that bacteria are connected with each other by appendages and also attach to PET film by appendages. The PET film can be almost completely degraded after 6 weeks at 30°C.
What does Ideonella sakaiensis look like?
Ideonella sakaiensis is Gram-negative, aerobic, and rod-shaped. The cells are motile with a polar flagellum. It grew within the pH range 5.5-9.0 (optimally at pH 7-7.5) and at 15-42 ºC (optimally at 30-37 ºC). Ideonella sakaiensis can grow on PET surfaces by adhering to the PET film and individual Ideonella sakaiensis using their thin appendages. These appendages may function to deliver PET-degrading enzymes to the PET film.
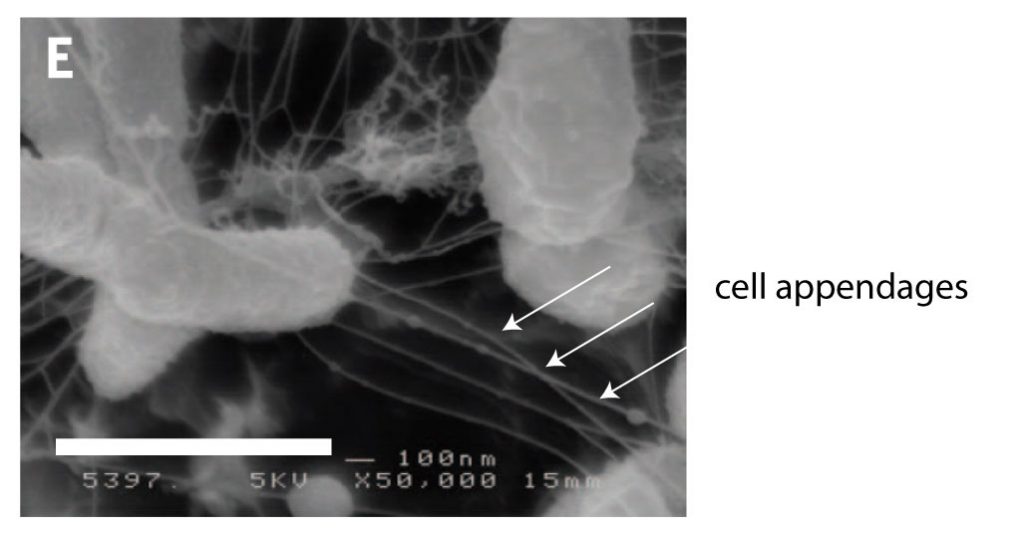
[In this image] Scanning electron microscopy image of Ideonella sakaiensis cells grown on PET film for 60 hours.
The arrows indicate cell appendages between cells. The cell appendages are long thin tubes extending from cells, and they connect to PET film surface and between cells. Scale bars, 1 mm. Magnification is 50,000x.
Photo source: Science. 2016 Mar 11;351(6278):1196-9. doi: 10.1126/science.aad6359.
How plastic-eating bacteria actually work?
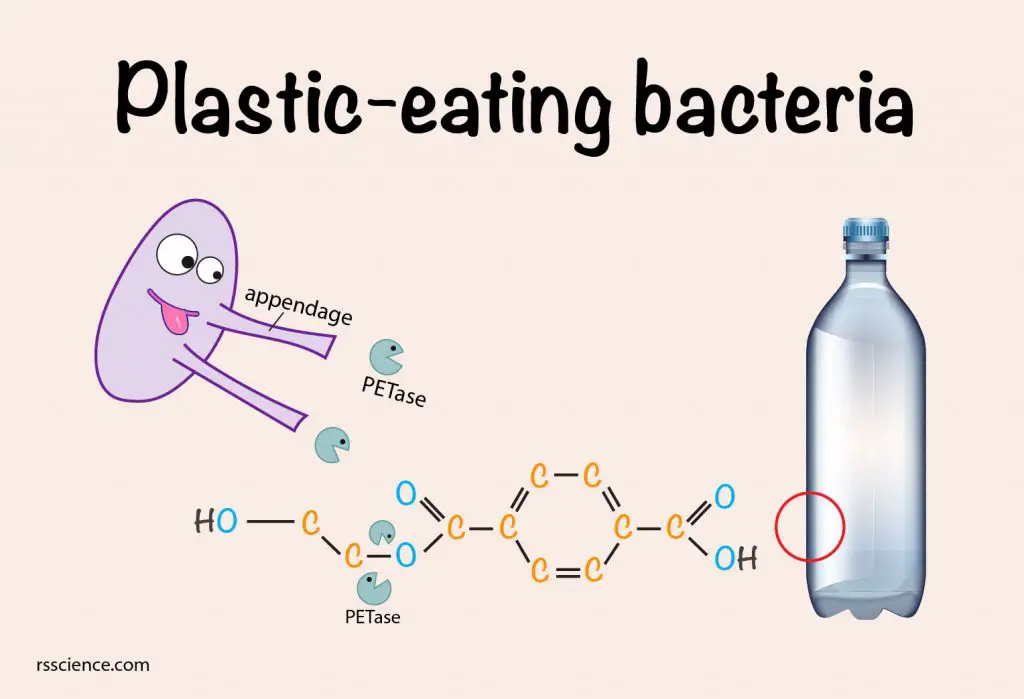
Many bacteria release enzymes to break down the nutrients that are too large to ingest. The newly discovered bacterium, Ideonella sakaiensis, applied the same strategy. I sakaiensis secretes enzymes called PETase that can break down PET into its most basic units.
PET is a long, repeating chain of chemicals. PETase secreted by I. sakaiensis breaks PET chains down to their individual units, Mono(2-hydroxyethyl) terephthalic acid (MHET), which is a heterodimer composed of terephthalic acid (TPA) and ethylene glycol (EG). Terephthalic acid can be imported into the I. sakaiensis for oxidation and incorporate into other metabolic pathways, whereas ethylene glycol is ready to use by I. sakaiensis and many other bacteria. As a result, both of the molecules derived from the PET are used by the cell to produce energy and to build necessary biomolecules. Eventually, they can convert plastic into CO2 and water.
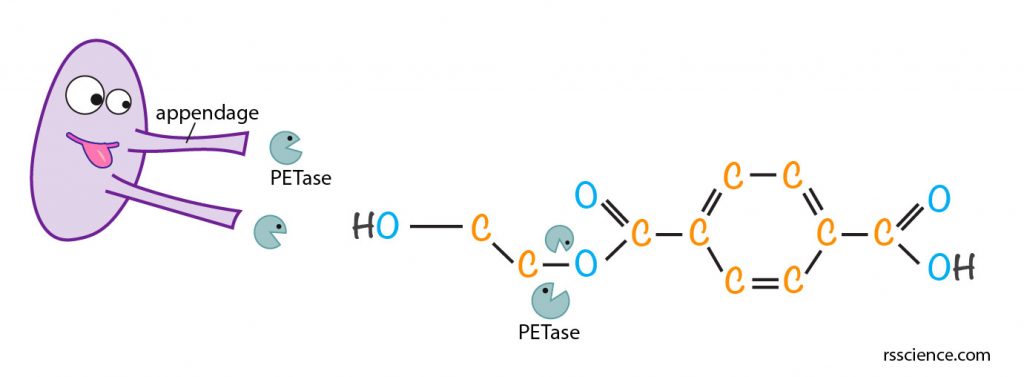
[In this figure] The illustration of how plastic-eating bacteria, I. sakaiensis, breaking down PET.
The plastic-eating bacteria, I. sakaiensis, secrete PETase through their long appendages when they grow on PET film. PETase is an enzyme that can break down PET long polymers into simple monomers by breaking down the C-O bond.
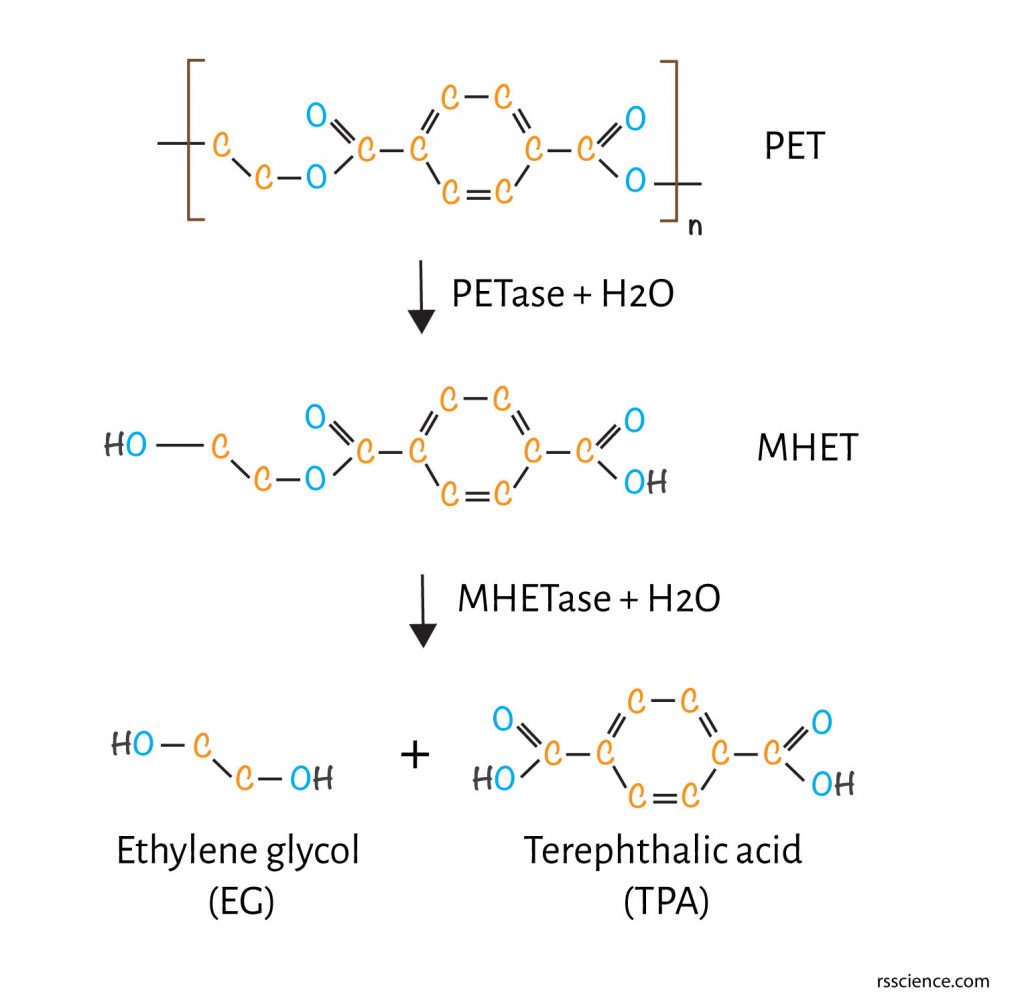
[In this figure] The PET degradation pathways.
Extracellular PETase hydrolyzes PET to produce MHET. MHETase, a predicted lipoprotein, hydrolyzes MHET to TPA and EG. TPA and EG can be used by I. sakaiensis and other microbes for making energy. Alternatively, the resulting monomers can be used for a new round of PET synthesis.
The improved version of PETase
So, are PETase and MHETase from the plastic-eating bacteria our hope for plastic pollution?
In my opinion, their discovery is impressive and is a good start to tackle the plastic pollution biodegradation. Although these enzymes are slow and inefficient, I believe that scientists can engineer a better version of enzymes with higher efficiency and specificity.
In 2018, a study published in the journal Proceedings of the National Academy of Sciences addressed how PETase works by determining the precise structure of the PETase enzyme. What they found is that the structure of PETase shares some common features of lipase and cutinase. Lipases are enzymes that break down lipids in our body. Cutinases break down cutin, one of two waxy polymers that are the main components of the plant cuticle. Making PETase more cutinase-like by two mutations on the active site of the PETase, they surprisingly improved PETase degradation capability.
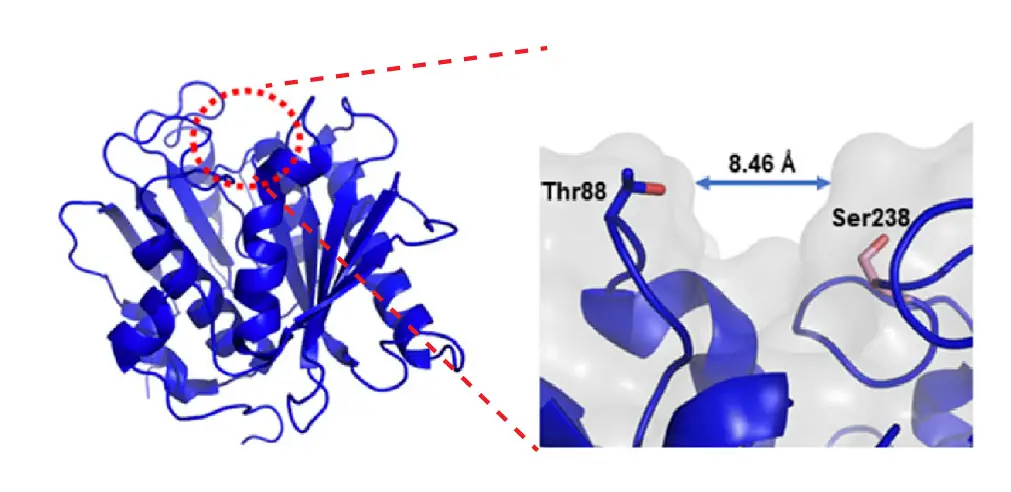
[In this figure] Cartoon representation of the PETase structure at 0.92 Å resolution.
The dashed red circle on the top indicates the active site of the PETase from I. sakaiensis. The right image is an enlarged view of the red dashed circle. Shortening the distance between Thr88 and Ser238 by mutation make it more cutinase-like, and surprisingly improved PETase degradation.
In the original report describing the discovery of PETase, Kohei Oda and Kenji Miyamoto’s team examined PETase digestions on PET films with a crystallinity of 1.9%, which is lower than that of most PET samples in either environment or in an industrial recycling plant. The improved PETase is active on PET of ~14.8% crystallinity (for reference, a commercial soft drink bottle is ~ 15.7%).
PET are polymers that contain ordered crystalline regions and disordered amorphous regions, see the figure below. The more well-ordered a polymer’s molecular chains are arranged, measured by a property called crystallinity, the harder they are for enzymes and organisms to break down.
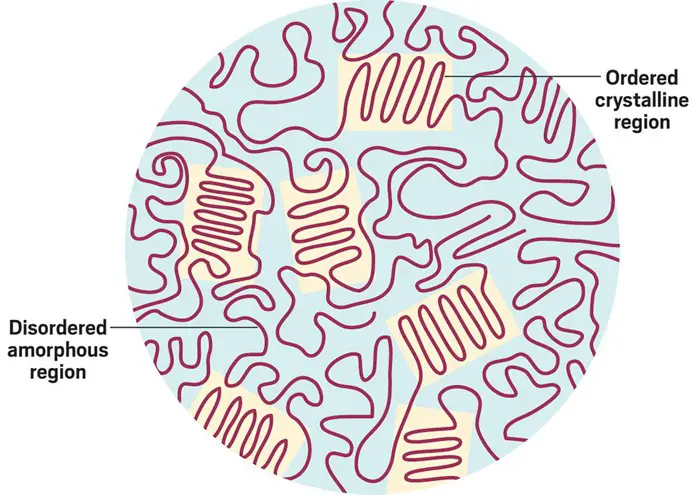
[In this figure] Crystallinity refers to the degree of structurally ordered. Amorphous refers to disordered regions in the polymer.
The complete degradation of PET is the ultimate goal, either to CO2 or recovered monomers for making fresh PET. The complete degradation needs to be carefully validated because there is a chance that leftover plastic fragments could harm the environment. Actually, the microplastic particle (small plastic particles) harm aquatic life more seriously than the bigger ones because they can eat them mistakenly and continually contaminate the food chain. People are now known to breathe microplastic particles, too.
Reference:
A bacterium that degrades and assimilates poly(ethylene terephthalate)
Ideonella sakaiensis sp. nov., isolated from a microbial consortium that degrades poly(ethylene terephthalate)
Structure of the plastic-degrading Ideonella sakaiensis MHETase bound to a substrate
Are Plastic-eating Enzymes Our Planet’s Only Hope?
Characterization and engineering of a plastic-degrading aromatic polyesterase