This article covers
What are peroxisomes? A quick overview
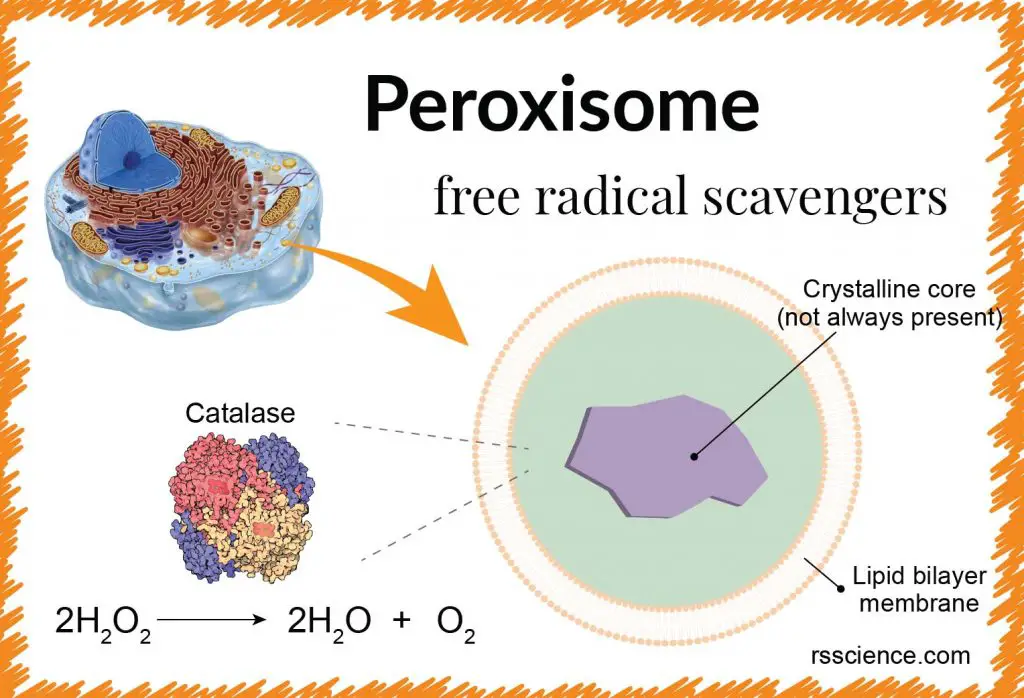
The peroxisome is a spherical membrane-bound organelle responsible for the fatty acid (oil molecule) breakdown in order to generate energy. Peroxisome also mediates many more biochemical reactions in virtually all eukaryotic cells.
Many enzymes inside the peroxisomes catalyze Redox (reduction-oxidation) reactions, which will generate hydrogen peroxide (H2O2) as a dangerous byproduct. A peroxisomal enzyme, called “Catalase”, can convert H2O2 into water (H2O) and oxygen (O2) to keep the cell safe. Peroxisomes owe their name to hydrogen peroxide generating and scavenging activities.
Peroxisomes – What does it look like?
Peroxisomes are small (0.1-1 µm diameter) organelles and located in the cytoplasm of a cell. Peroxisomes are too small to be seen under a regular light microscope. Electron microscope or fluorescence microscope are required to observe and study peroxisomes.
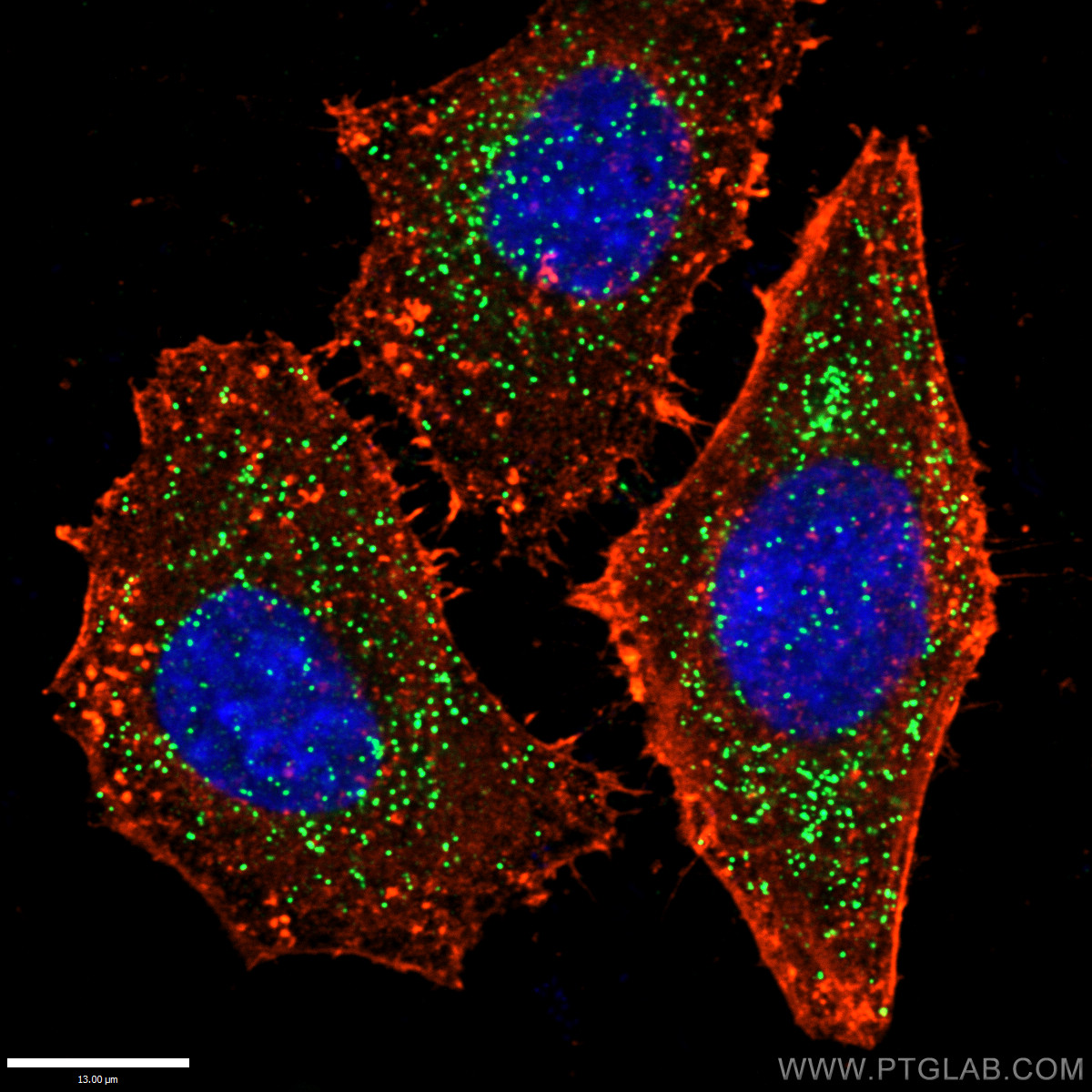
[In this figure] Immunofluorescent image of peroxisomes (green) by an antibody against a peroxisomal membrane protein, PEX14 (peroxisomal biogenesis factor 14).
Photo source: https://www.ptgcn.com/products/PEX14-Antibody-10594-1-AP.htm
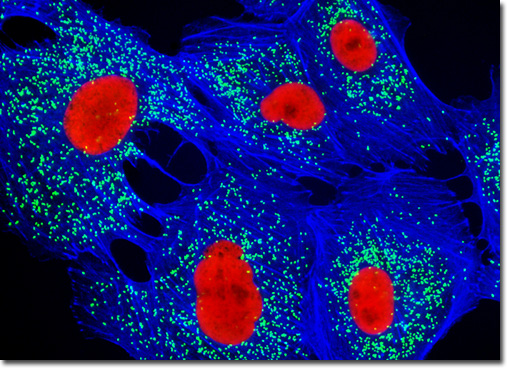
[In this figure] Immunofluorescent image of peroxisomal membrane protein 70 (PMP 70, cyan); nuclei (red); membrane (blue).
Photo source: Michael W. Davidson
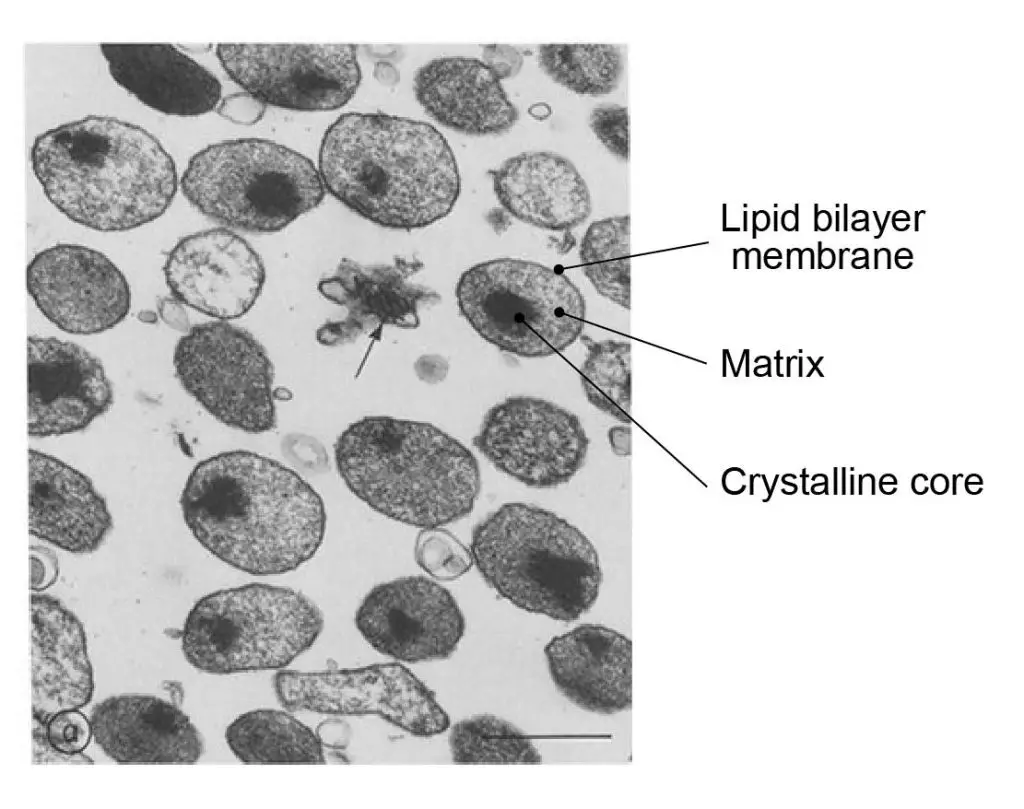
[In this figure] Electron micrographs of purified peroxisomes. Magnification 38,000x.
Photo source: Fujiki Y et al., J Cell Biol. 1982
The structure of peroxisome
The peroxisome is a membrane-bound vesicle containing a fine, granular matrix. Peroxisome’s membrane is similar to the cell membrane or ER. The space compartmentalized by the membrane creates an optimized environment to promote various metabolic reactions that are required to sustain cellular functions and the viability of the organism.
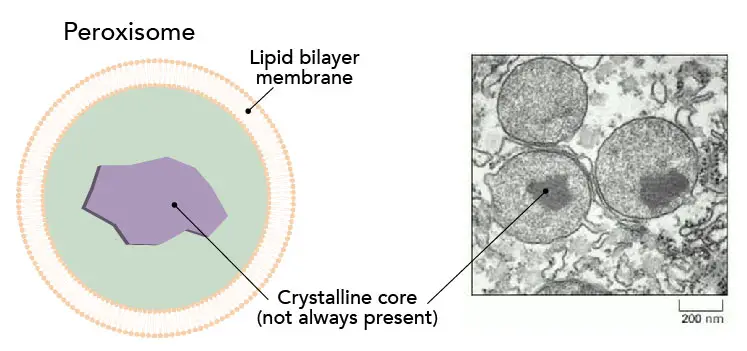
[In this figure] Peroxisomes.
Left: The structure of the peroxisome. They have a simple structure consisting of two major parts; an outer layer, a single lipid-bilayer membrane, and a crystalline core.
Right: an electron microscopy image of peroxisomes. In this example, the peroxisomes contain a highly condensed urate oxidase crystalline core. The enzymes are accumulated in large numbers so once they are needed in the cell, they can be released quickly so the peroxisomes can act rapidly when required. (Image from Schrader, M. and Fahimi, H. 2008. The peroxisome: still a mysterious organelle. Histochemistry and Cell Biology 129(4), pp. 421-440.)
Sometimes, a condensed crystalline core can be seen in the center of the peroxisome. The core contains a variety of enzymes for the catalysis of cell functions, mainly metabolism. Typically, the most concentrated of these enzymes, catalase or urate oxidase, can become so high in number that they aggregate to form a crystalline core in the center of the organelle. This structure can be seen in electron micrographs.
The number, size, and protein composition of peroxisomes are variable and depend on cell type and environmental conditions. For example, in baker’s yeast (S. cerevisiae), only a few, small peroxisomes are present if the cells have an abundant glucose supply. In contrast, when the yeasts were supplied with long-chain fatty acids as the sole nutrient source, cells generate more large peroxisomes to meet the metabolic demand.
[In this video] Time-lapse microscopy of mitochondria labeled with red and peroxisomes labeled with green. The two organelles show complex interactions. Video plays at 48x real-time.
What does peroxisome do?
Peroxisome is a multifunctional biochemistry laboratory in the cell
Peroxisomes contain more than 50 different enzymes, which are involved in a variety of biochemical reactions. Peroxisomes originally were defined as organelles that carry out oxidation reactions leading to the production of hydrogen peroxide (H2O2). Because hydrogen peroxide is harmful to the cell, peroxisomes also contain the enzyme catalase, which decomposes hydrogen peroxide by converting it to water. A variety of substrates are broken down by such oxidative reactions in peroxisomes, including uric acid, amino acids, and fatty acids. The oxidation of fatty acids is a particularly important example since it provides a major source of metabolic energy.
The chemical reactions are potentially dangerous to the cells. This is why we need the peroxisomes to control chemical reactions within a membrane-bound space separated from the rest of the cells. Let me here pay tribute to those peroxisomes which handle the high-risk tasks for us.
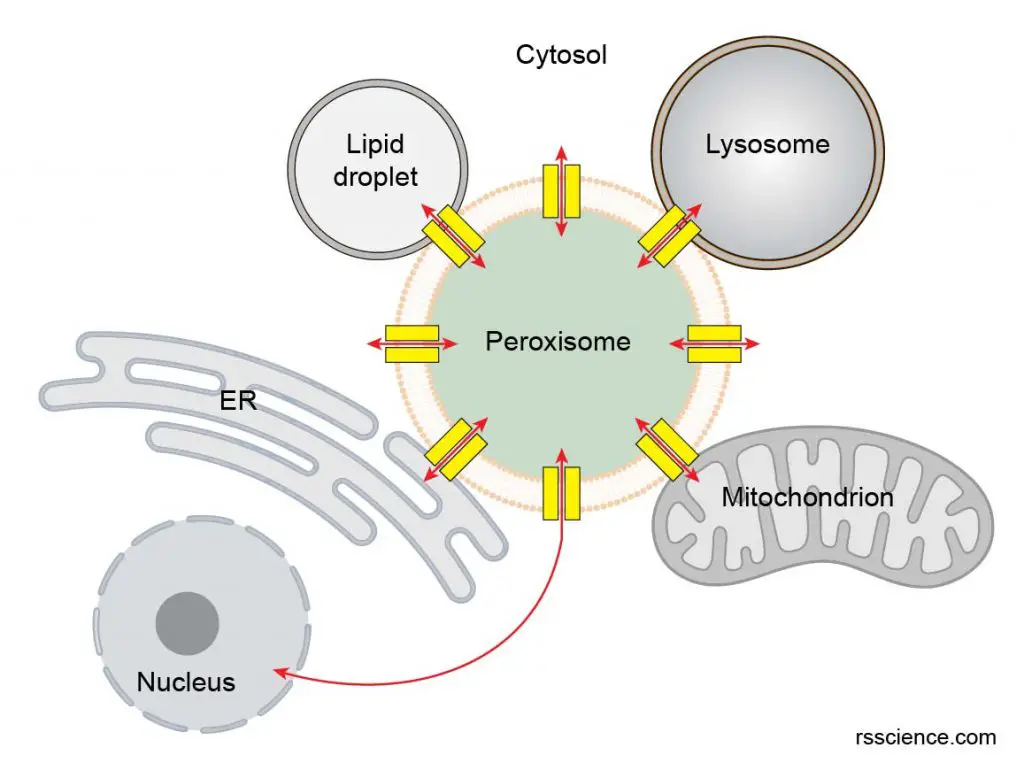
[In this figure] Peroxisomes closely interact with other organelles in the cells. The biomolecules are transported into peroxisomes for specific chemical reactions. The products are also exported to other organelles for biological functions.
Peroxisomal enzymes mediate Redox reactions and generate hydrogen peroxide
From the chemical point of view, many enzymes inside the peroxisomes catalyze Redox (reduction-oxidation) reactions.
Oxygen is an active molecule, and many chemical reactions happen base on the reactivities of oxygen. Generally speaking, oxidation is adding an oxygen (increase in the oxidation state), and reduction is removing an oxygen (decrease in the oxidation state) from a molecule. Typically, oxidation is burning. When a piece of wood on the fire, the oxidation state of carbon atoms in the wood increase by the reaction of oxygen gas, turning the carbon into carbon dioxide (CO2).
When a reductive reaction happens in the peroxisome, the enzyme takes away oxygen (in the form of superoxide O2•−). However, the enzyme cannot hold the oxygen forever, so the oxygen is transferred to a water molecule. As a result, the water molecule is oxidized to become a hydrogen peroxide (H2O2).

[In this figure] A stable atom has a balanced number of electrons, no more, no less. Free radicals eagerly want to steal electrons from other atoms to fulfill their unstable status. Antioxidants have free electrons that can give to free radicals to calm them down.
The molecules that contained chemically reactive oxygen (like O2•−, •OH, H2O2, and NO) are called reactive oxygen species (ROS) or free radicals. Many ROS are created as an unavoidable byproduct of normal cellular metabolism. These ROS need to be removed from the cells carefully; otherwise, ROS will damage the cells by unwanted reactions with DNA, lipid, and proteins.
In fact, many diseases like cancers and aging originate from the bad effects of ROS in our bodies. Radiation, tobacco, and drugs also increase the chances of damage by ROS. Antioxidants or free-radical scavengers can cancel out the effects of ROS. This is why we are encouraged to eat more healthy food that enriches natural antioxidants, like vitamins A, C, and E.
Peroxisome and catalase – free radical scavengers
As we mention, the peroxisomes generate many hydrogen peroxide molecules (H2O2) as dangerous byproducts. In fact, this is why the “peroxi”-some got its name. Fortunately, the peroxisomes are able to contain these H2O2 and break them down into water (H2O) and oxygen (O2). This critical reaction is done by another peroxisomal enzyme, called “Catalase”.
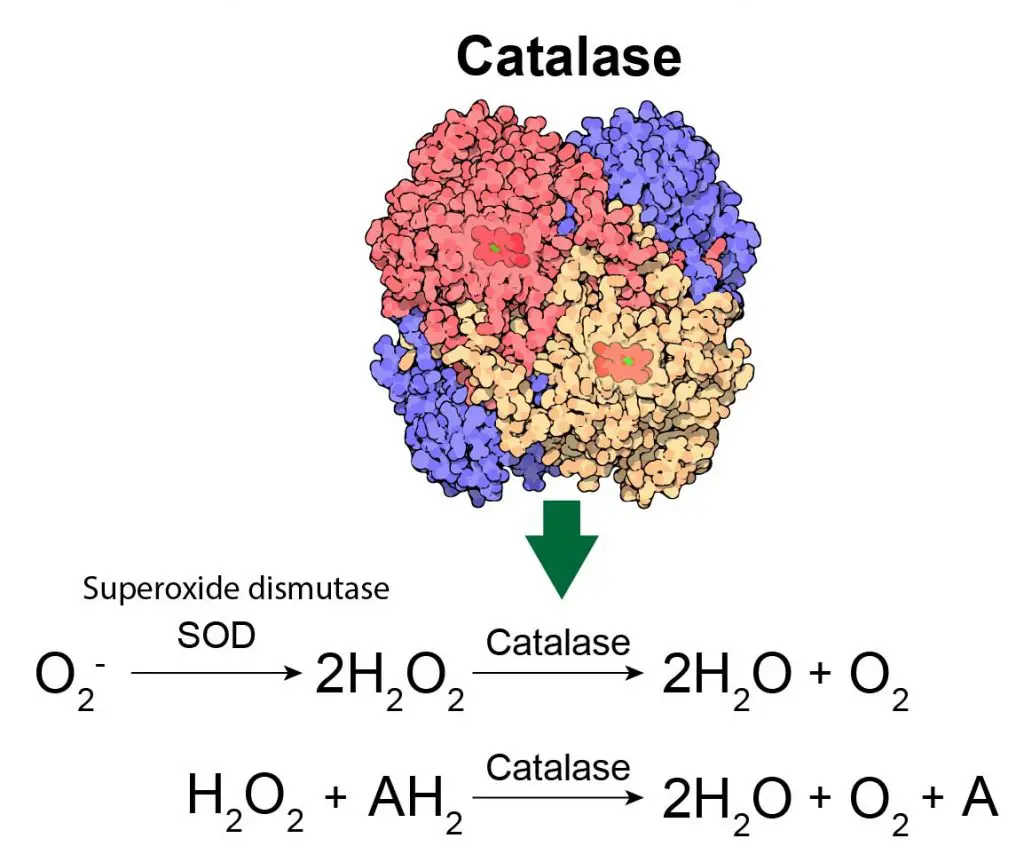
[In this figure] Catalase is an enzyme that catalyzes the degradation of hydrogen peroxide by converting it to water and oxygen. Alternatively, catalase can use hydrogen peroxide to catalyze the oxidation of an organic molecule (A), which results in the conversion of hydrogen peroxide to water.
Peroxisome helps the breakdown of fatty acids
Fatty acids are the basic units of fat and oil. The foods we eat include a variety of fatty acids. For example, fatty acids could be either saturated or unsaturated fats. They can be further classified according to their different lengths of carbon chains (from 4 to 28 carbon atoms).
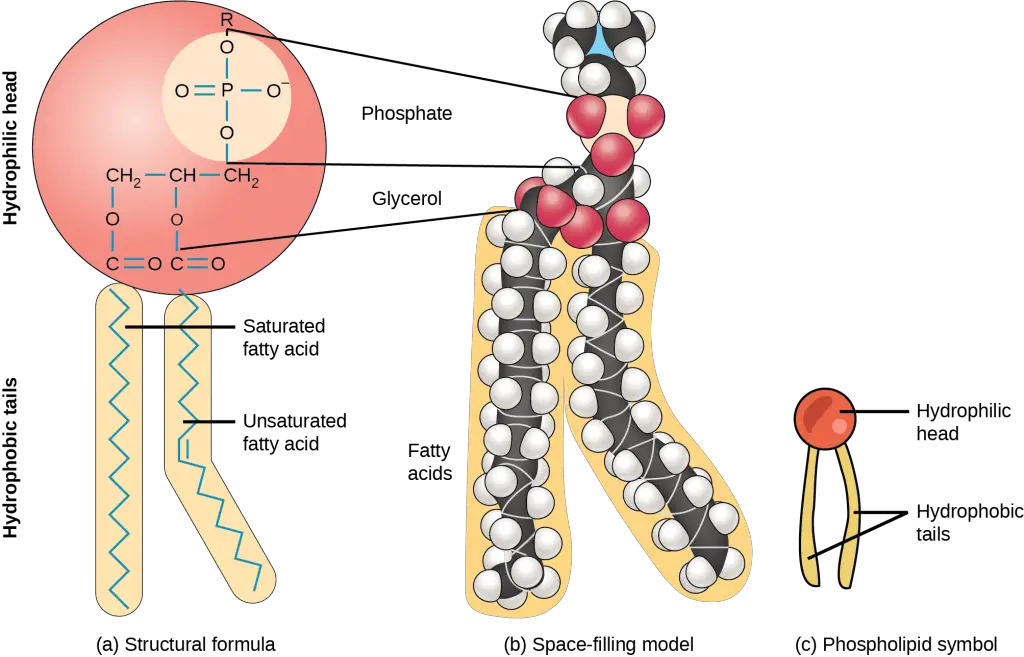
[In this figure] A lipid molecule with two fatty acid chains (18 carbon atoms). One chain is saturated, and the other one is unsaturated.
To use these fatty acids, the peroxisomes have to breakdown some of the long and branched-chain fatty acids into simpler nutrients. This process involves several special enzymes and oxidative reactions (alpha- and beta-oxidation) accompanied by the production of hydrogen peroxide (H2O2) from oxygen.
More biochemical reactions in peroxisomes
In addition to providing a compartment for oxidation reactions, peroxisomes are involved in lipid biosynthesis. In animal cells, cholesterol and dolichol are synthesized in peroxisomes as well as in the ER. In the liver, peroxisomes are also involved in the synthesis of bile acids, which are derived from cholesterol.
Besides, peroxisomes contain enzymes required for the synthesis of plasmalogens—a family of phospholipids in which one of the hydrocarbon chains is joined to glycerol by an ether bond rather than an ester bond. Plasmalogens are important membrane components in some tissues, particularly the heart and brain. In addition, the peroxisomes in the liver cells also handle the detoxifications of many chemicals, including alcohol and drugs. It is fair to say that peroxisome is the chemistry laboratory inside the cell.
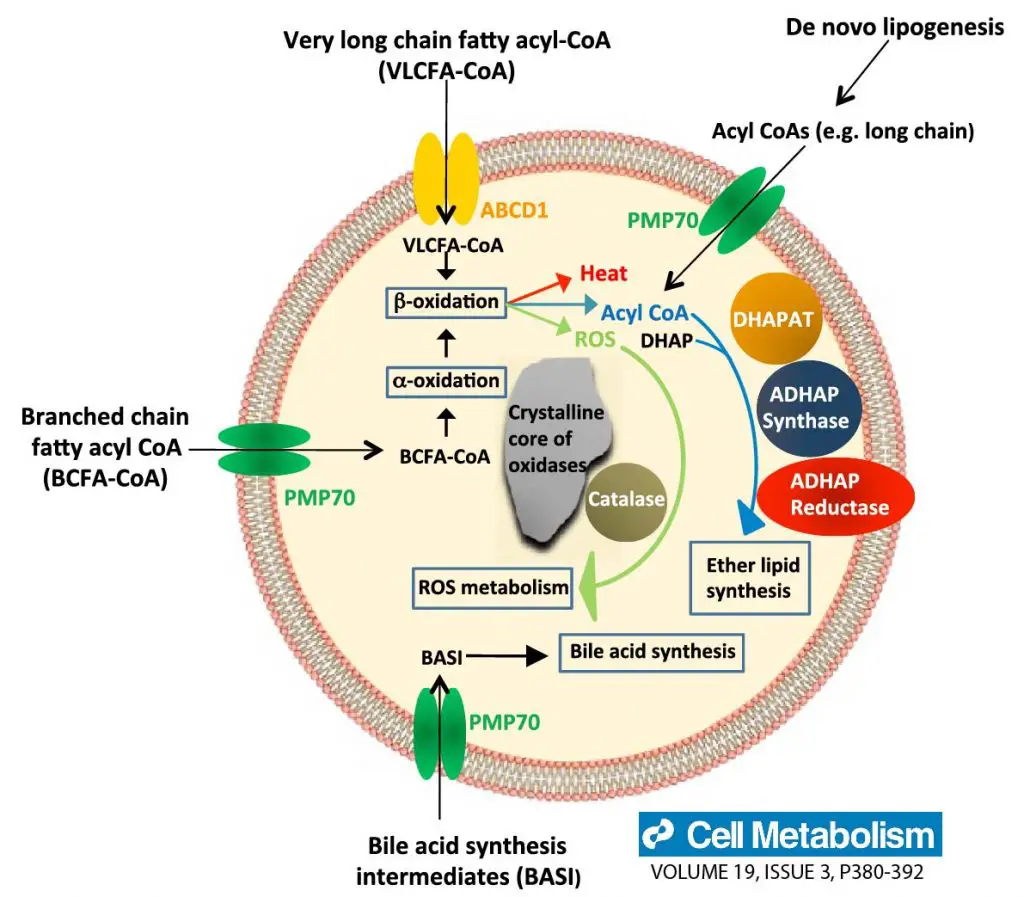
[In this figure] Summary of peroxisome functions.
The main metabolic functions of peroxisomes in mammalian cells include β-oxidation of very long-chain fatty acids, α-oxidation of branched-chain fatty acids, synthesis of bile acids, and ether-linked phospholipids, and removal of reactive oxygen species.
Do plant cells have peroxisome?
Yes, peroxisomes can be found in the cytoplasm of virtually all eukaryotic cells including both animal and plant cells.
In plant cells, peroxisomes carry out two additional important roles.
First, peroxisomes (also called glyoxysomes) in seeds are responsible for the conversion of stored fatty acids to carbohydrates, which is critical to providing energy and raw materials for the growth of the germinating plant. This occurs via a series of reactions termed the glyoxylate cycle.
Second, peroxisomes in leaves are involved in the recycling of carbon from phosphoglycolate (a side product formed during photosynthesis) during photorespiration.
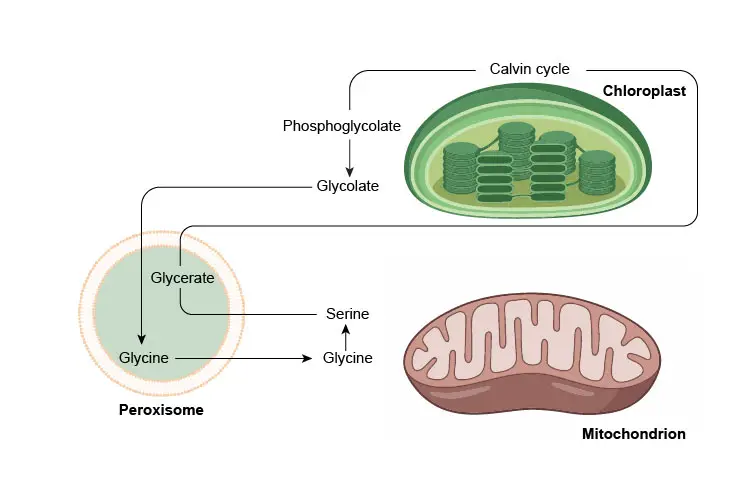
[In this figure] Photorespiration involves a complex network of enzyme reactions that exchange metabolites between chloroplasts, leaf peroxisomes, and mitochondria.
Peroxisome assembly and biogenesis
The assembly of peroxisomes is fundamentally similar to that of mitochondria and chloroplasts, rather than to that of the endoplasmic reticulum, Golgi apparatus, and lysosomes. Proteins are synthesized on free cytosolic ribosomes and then imported into peroxisomes. Phospholipids are also imported from the smooth endoplasmic reticulum to peroxisomes. The import of proteins and phospholipids results in peroxisome growth, and new peroxisomes are then formed by the division of old ones.
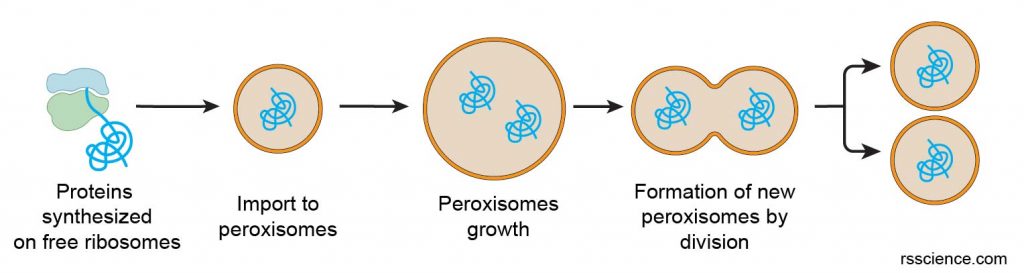
[In this figure] Assembly of peroxisomes.
Proteins destined for peroxisomes are synthesized on free ribosomes. Protein import results in peroxisome growth. The formation of new peroxisomes by the division of old ones.
The discovery of peroxisome
Peroxisomes were identified as organelles by the Belgian cytologist Christian de Duve in 1967. De Duve and co-workers discovered that peroxisomes contain several oxidases involved in the production of hydrogen peroxide (H2O2) as well as catalase involved in the decomposition of H2O2 to oxygen and water. Due to their role in peroxide metabolism, De Duve named them “peroxisomes”, replacing the formerly used morphological term “microbodies”.

[In this figure] The Nobel Prize in Physiology or Medicine 1974 was awarded jointly to Albert Claude, Christian de Duve, and George E. Palade for their works in cell biology, especially for the discoveries of cell organelles, lysosome, peroxisome, and ribosome.
Photo source: The Nobel Prize.
Lysosome vs Peroxisome
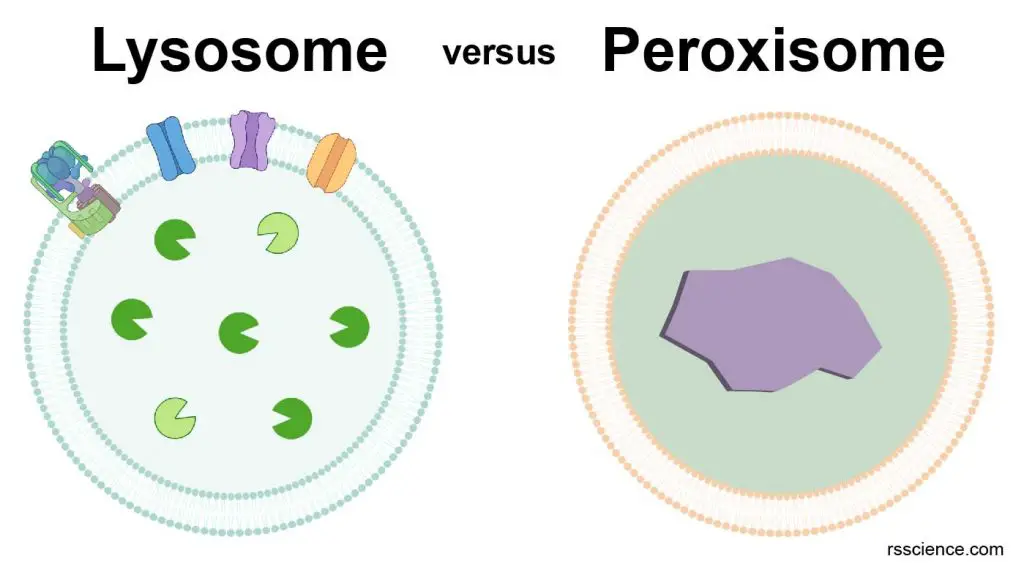
Lysosome and peroxisome are two cell organelles often compared. We made a table here to summarize the difference.
| Lysosome | Peroxisome |
| Lysosomes break down biological polymers like proteins (to amino acid) and polysaccharides (to sugar) | Peroxisomes oxidize organic compounds, break down fatty acids, and degrade hydrogen peroxides. |
| Cell’s recycling center | Cell’s chemical laboratory |
| Consists of degradative enzymes | Consists of oxidative enzymes |
| Responsible for the digestion (materials from outside or inside the cells) | Responsible for the protection of the cells from reactive oxygen species or free radicals |
| Found in animals (vacuoles in plant) | Found in all eukaryotes |
| Derived from the fusion of endosomes and Golgi apparatus-derived vesicles | Derived from endoplasmic reticulum and are capable of replicating by themselves |
| Lysosomal enzymes produced in rough endoplasmic reticulum | Peroxisomal enzymes produced on free ribosomes |
| Comparatively large in size; various by cell types; 0.5-1.0 µm | Comparatively small; 0.2-1.0 µm |
| Involved in endocytosis, phagocytosis and autophagy | Involved in biosynthesis of lipids and bile acids; photorespiration in plant cells |
| Degradative reactions do not generate energy | Oxidative reactions in peroxisomes break down fatty acids for ATP generation |
Peroxisomal disorders
Peroxisomal disorders are diseases caused by mutations in genes that are involved in peroxisome biogenesis. Peroxisomal disorders are congenital disorders, and they range from relatively moderate to severe in nature. For example, Zellweger syndrome is characterized by complete absence or reduction in the number of peroxisomes. Mutations giving rise to Zellweger syndrome cause copper, iron, and very long-chain fatty acids to accumulate in the blood and in tissues, such as the liver, brain, and kidneys. Infants with Zellweger syndrome are often born with a facial deformity and intellectual disability. Most infants with Zellweger syndrome do not live beyond one year.
Summary
- Peroxisome is a spherical organelle responsible for the fatty acid (oil molecule) breakdown in order to generate energy.
- Peroxisomes in the liver cells also handle the detoxification of many chemicals, including alcohol and drugs.
- Many enzymes inside the peroxisomes catalyze Redox (reduction-oxidation) reactions, which will generate hydrogen peroxide (H2O2) as a dangerous byproduct.
- Peroxisomal enzyme, called “Catalase”, can convert H2O2 into water (H2O) and oxygen (O2) to keep the cell safe.
- The main metabolic functions of peroxisomes in mammalian cells include β-oxidation of very long-chain fatty acids, α-oxidation of branched-chain fatty acids, synthesis of bile acids, and ether-linked phospholipids, and removal of reactive oxygen species.
References
“Peroxisomes” The Cell: A Molecular Approach. 2nd edition.
“Difference Between Lysosome and Peroxisome”
“Peroxisomes: A Nexus for Lipid Metabolism and Cellular Signaling”